The first documented case of a person contracting HIV while taking pre-exposure prophylaxis (PrEP) has been presented at the Retroviruses and Opportunistic Infections Conference in Boston overnight.
Evidence presented by David Knox, MD, an HIV specialist at the Maple Leaf Medical Clinic, suggested that a 43-year-old man who has sex with men and had been adhering well to PrEP over a 24 month period has tested positive for HIV.
Initial tests indicated that he was acutely (very recently) infected: He tested positive for the p24 antigen, which appears within about three weeks of HIV infection and disappears a few weeks afterward; and at that time he tested negative for HIV antibodies, which typically appear two to eight weeks after infection.
Head of the first approved PrEP trial in San Francisco in 2010 Robert Grant MD said it shows that nothing is 100% safe.
“After 32 years of experience with HIV research, I have learned never to say ‘never’,” said Robert M. Grant, MD, MPH, a professor at the University of California, San Francisco, who was the head of the iPrEx trial that first proved PrEP’s effectiveness among MSM and transgender women in 2010.
“Yet I also think that gay men benefit from feeling safer during sex, and I am grateful that PrEP affords that feeling.”

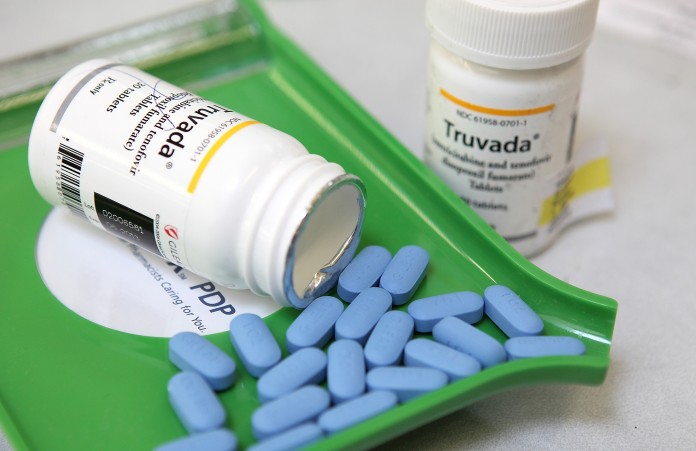
Pharmacy records indicated that the man in the case study had consistently filled his Truvada prescription on schedule.
Dried blood spot testing on a sample taken 16 days after he tested positive for HIV indicated that he had adhered well to Truvada during the previous one to two months, a period that overlapped with the estimated time when he contracted the virus.
“This person claims he was taking PrEP every day and I believe him,” said Grant.
The man tested positive for a strain of the virus that was resistant to multiple drugs, including emtricitabine and tenofovir, which make up Gilead’s Truvada, the current antiretroviral cocktail on the market that’s used as PrEP.
“Despite all these resistance mutations, the man in the case study is currently taking HIV treatment and has a fully suppressed viral load.”
What is rarer is a virus that is highly resistant to both tenofovir and emtricitabine, as in this new case report.
Indeed, according to Grant, among more than 9,200 participants in the clinical trials of PrEP, such a virus that was highly resistant to both components of Truvada was never seen.
In Australia, some states are currently undertaking further PrEP trials to test its effectiveness and the limited availability of PrEP. As yet New Zealand hasn’t made any inroads but it is something the New Zealand AIDS Foundation are monitoring closely.
Last Updated on Feb 26, 2016
The news team for Gay Nation love tips from our readers. Got tips or a news story that you would like published? Go here to tell us something.
Visit the Gay Nation store Now